Advancing ophthalmology: Unither invests in its French R&D site.
Carragelose® Eyedrops
About Unither Pharmaceuticals
Unither Pharmaceuticals is a pharmaceutical contract manufacturer specialized in sterile and non-sterile liquid forms, including single-doses, multi-doses, vials, liquid stick packs, and sprays.
In the pharmaceutical contract manufacturing sector, the company specializes in particular in the manufacturing of sterile single doses using Blow-Fill-Seal (BFS) technology, of which it is the world leader, with an annual production capacity exceeding 5 billion doses. Unither develops and manufactures exclusive turnkey products for its clients, marketed under their own brand, and supports their industrial strategies on an international scale.
Key expertise
As a Contract Development and Manufacturing Organization (CDMO), we cover all services aimed at producing healthcare products, from pre-clinical phases to commercial production for our pharmaceutical industry clients, whether large companies or start-ups. We are proud of our co-development method, which is based on close collaboration with our clients to bring their innovative health solutions to life. Our expertise extends beyond manufacturing to offer turnkey solutions, including the development of comprehensive product documentation ready for licensing.
Manufacturing
Development
Out-licensing
Co-Development Program
Regulatory Support
Areas of application
Key figures
9
production sites
In Europe, the United States, Brazil, and China
5 billion
doses produced per year
World leader in Blow-Fill-Seal (BFS) technology
+ 100
distribution countries
Solutions tailored to your markets
+2500
employees
Committed to our customers
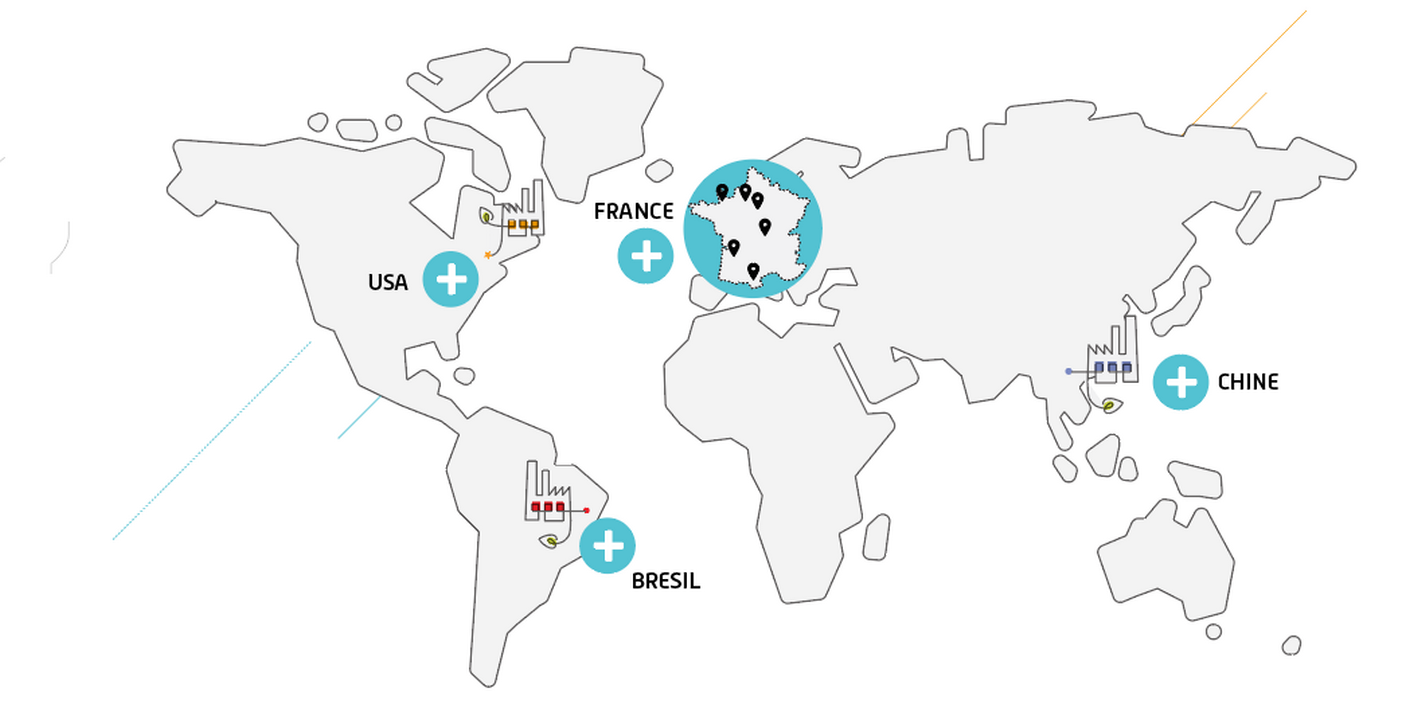
International presence
We operate on four continents (Europe, North America, South America, and Asia) through 8 industrial sites, as well as in our Innovation and Development center and our commercial offices in Paris, Brazil, and China. We are continuing to grow internationally, strengthening our capacities and our ability to better meet our customers’ development and manufacturing needs.
Quality & regulation
Unither Pharmaceuticals operates in strict compliance with international regulatory requirements.
Its sites are inspected and approved by leading health authorities (including the EMA, ANSM, FDA, ANVISA, MHRA, Health Canada, TGA, MFDS, and local authorities, and operate in compliance with Good Manufacturing Practices (cGMP / GMP).”
Unither is also certified in ISO 13485:2016 for medical devices and holds site-specific certifications (including in ISO 50001 for energy management) and abides by ICH and WHO guidelines for stability studies. Several sites also benefit from the Research Tax Credit (CIR).
Come and meet us
View AllFAQ
Unither Pharmaceuticals is the world leader in Blow-Fill-Seal (BFS) technology and a specialist in sterile liquid formats. Our presence in Europe, North and South America, and Asia ensures volume security, supply continuity, and proximity to your markets.
With robust cGMP processes, expertise in international regulations, and an efficient co-development model, Unither accelerates and supports your projects, from formulation to commercial manufacturing.
Unither offers several flexible collaboration models, tailored to the needs of pharmaceutical industry players, from innovative start-ups to large international pharmaceutical companies.
Collaboration models tailored to your projects
Depending on your objectives, Unither offers different types of partnerships:
- Contract manufacturing: production of clinical and commercial batches at industrial scale
- Licensing-out: turnkey product solutions designed to meet patient and market needs
- Co-development: close collaboration to design, develop, and industrialize a new product
We produce sterile and non-sterile liquid medicines and medical devices, as well as single-dose products, sprays, stick-packs, vials, and tablets.


